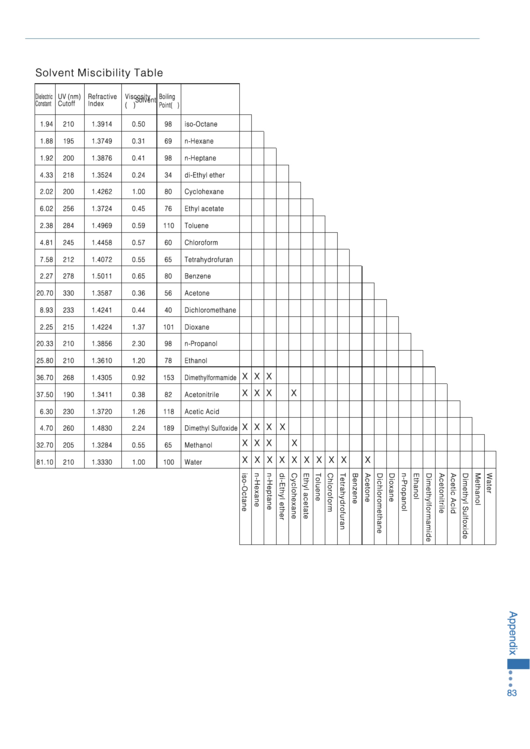
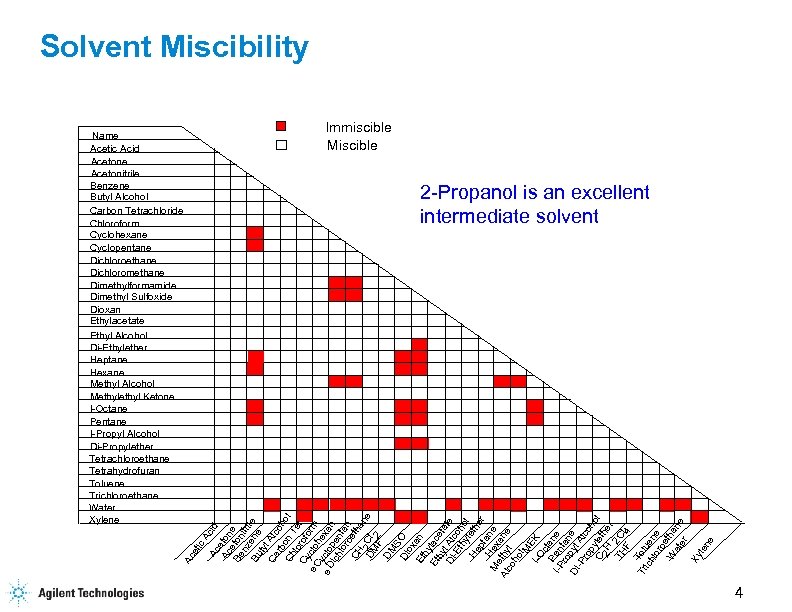
Miscibility Chart Solvents
Miscibility Chart Solvents - Web a solvent miscibility chart is used to determine the compatibility of different solvents, or substances that dissolve other substances, to mix together effectively. Web draw one solute molecule interacting with several solvent molecules. The following compounds are liquid at room temperature and are completely miscible with water; Miscibility describes the behavior of solutions that are mixtures of 2 or more liquids. Many of them are hygroscopic. The document lists various chemical compounds and provides their polarity index, viscosity, uv cutoff wavelength, and solubility in water. It helps in understanding which solvents can be mixed without forming unwanted precipitates or separating into layers. Label the strongest intermolecular force holding them together. They are often used as solvents. Web twenty solvents are available, most of which can also be purchased stored over molecular sieves. The document lists various solvents and provides their polarity index, viscosity, uv cutoff wavelength, solubility in water, and miscibility. Predict whether the solvent will dissolve significant amounts of the solute. Web substances are said to be miscible in one another if they dissolve to form a uniform solution. The following compounds are liquid at room temperature and are completely miscible with water; Web solvent miscibility table solvent polarity chart relative compound. Ethyl acetate heptane • hexane • • isooctane • isopropanol. In other words, “like dissolves like.” We often think of solutions as a combination of a liquid and a solid. Web use this handy chart to look up miscibility of common liquid solvents. All of the solvents are packaged under nitrogen in the acroseal septum sealed glass bottle to protect product integrity and maintain dryness. Predict whether the solvent will dissolve significant amounts of the solute. Miscibility describes the behavior of solutions that are mixtures of 2 or more liquids. It helps in understanding which solvents can be mixed without forming unwanted precipitates or separating into layers. Many of them are hygroscopic. Lc i gcs chromacademy crawford scientific eoooo acetic acid acetone acetonitrile 000000 benzene. Acetic acid acetone acetonitrile benzene. They are often used as solvents. Bookmark or download our miscibility table for common lab solvents. The following compounds are liquid at room temperature and are completely miscible with water; The document lists various chemical compounds and provides their polarity index, viscosity, uv cutoff wavelength, and solubility in water. Acetic acid acetone acetonitrile benzene. For reverse phase chromatography eluent strength decreases as its polarity increases 2 uv cutoff, the wavelength at which the solvent absorbance in Electrophoresis, western blotting and elisa In other words, “like dissolves like.” Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for identifying suitable mobile. Predict whether the solvent will dissolve significant amounts of the solute. Lc i gcs chromacademy crawford scientific eoooo acetic acid acetone acetonitrile 000000 benzene miscible mmiscible butanol carbon tetrachloride chloroform cyclohexane cyclopentane dichloroethane dichloromethane diethyl ether dimethylformamide dimethyl sulfoxide dioxane dipropyl ether ethanol ethyl acetate. Web sign up for email customer service +351 21 425 33 50 ; Acetic acid. Web sign up for email customer service +351 21 425 33 50 ; Bookmark or download our miscibility table for common lab solvents. Many of them are hygroscopic. The document lists various solvents and provides their polarity index, viscosity, uv cutoff wavelength, solubility in water, and miscibility. Web solvent miscibility table solvent polarity chart relative compound. Electrophoresis, western blotting and elisa All of the solvents are packaged under nitrogen in the acroseal septum sealed glass bottle to protect product integrity and maintain dryness. Web sign up for email customer service +351 21 425 33 50 ; Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for identifying. We often think of solutions as a combination of a liquid and a solid. Many of them are hygroscopic. In other words, “like dissolves like.” It displays solvents in columns and rows and indicates with symbols whether each solvent pair is miscible or immiscible. Web use this handy chart to look up miscibility of common liquid solvents. Web solvent miscibility table solvent polarity chart relative compound. Ethyl acetate heptane • hexane • • isooctane • isopropanol. Electrophoresis, western blotting and elisa Label the strongest intermolecular force holding them together. They are often used as solvents. Electrophoresis, western blotting and elisa Bookmark or download our miscibility table for common lab solvents. For reverse phase chromatography eluent strength decreases as its polarity increases 2 uv cutoff, the wavelength at which the solvent absorbance in Web sign up for email customer service +351 21 425 33 50 ; In other words, “like dissolves like.” Acetic acid acetone acetonitrile benzene. All of the solvents are packaged under nitrogen in the acroseal septum sealed glass bottle to protect product integrity and maintain dryness. Predict whether the solvent will dissolve significant amounts of the solute. Web use this handy chart to look up miscibility of common liquid solvents. Web twenty solvents are available, most of which can. Web sign up for email customer service +351 21 425 33 50 ; Web a solvent miscibility chart is used to determine the compatibility of different solvents, or substances that dissolve other substances, to mix together effectively. Many of them are hygroscopic. We often think of solutions as a combination of a liquid and a solid. Web antibodies & protein biology antibody production & purification; Lc i gcs chromacademy crawford scientific eoooo acetic acid acetone acetonitrile 000000 benzene miscible mmiscible butanol carbon tetrachloride chloroform cyclohexane cyclopentane dichloroethane dichloromethane diethyl ether dimethylformamide dimethyl sulfoxide dioxane dipropyl ether ethanol ethyl acetate. The polarity index increases with polarity. They are often used as solvents. Ethyl acetate heptane • hexane • • isooctane • isopropanol. For reverse phase chromatography eluent strength decreases as its polarity increases 2 uv cutoff, the wavelength at which the solvent absorbance in Label the strongest intermolecular force holding them together. Web draw one solute molecule interacting with several solvent molecules. Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for identifying suitable mobile phase solvents. In other words, “like dissolves like.” All of the solvents are packaged under nitrogen in the acroseal septum sealed glass bottle to protect product integrity and maintain dryness. Acetic acid acetone acetonitrile benzene.Top Solvent Miscibility Charts free to download in PDF format
Solvent Miscibility Chart Download Printable Pdf Templateroller Labb
Solvent Miscibility Chart Download Printable PDF Templateroller
Solubility Factors When Choosing a Solvent Labclinics
Solvent Miscibility Chart A Visual Reference of Charts Chart Master
Solvent Miscibility Chart Download Printable Pdf Templateroller Labb
Solvent Miscibility Table
Miscible Definition in Chemistry What Is Miscibility?
Solvent Miscibility
Solvent Miscibility Chart A Visual Reference of Charts Chart Master
It Helps In Understanding Which Solvents Can Be Mixed Without Forming Unwanted Precipitates Or Separating Into Layers.
Bookmark Or Download Our Miscibility Table For Common Lab Solvents.
All Of The Solvents Are Packaged Under Nitrogen In The Acroseal Septum Sealed Glass Bottle To Protect Product Integrity And Maintain Dryness.
Miscibility Describes The Behavior Of Solutions That Are Mixtures Of 2 Or More Liquids.
Related Post: