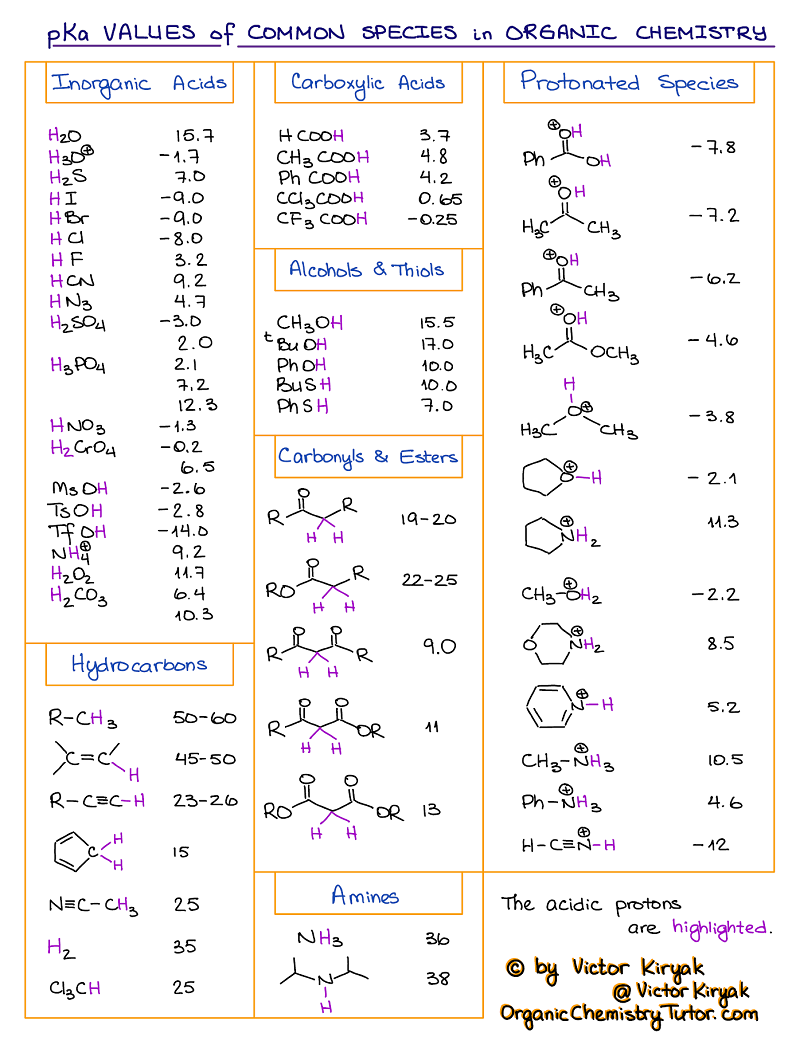
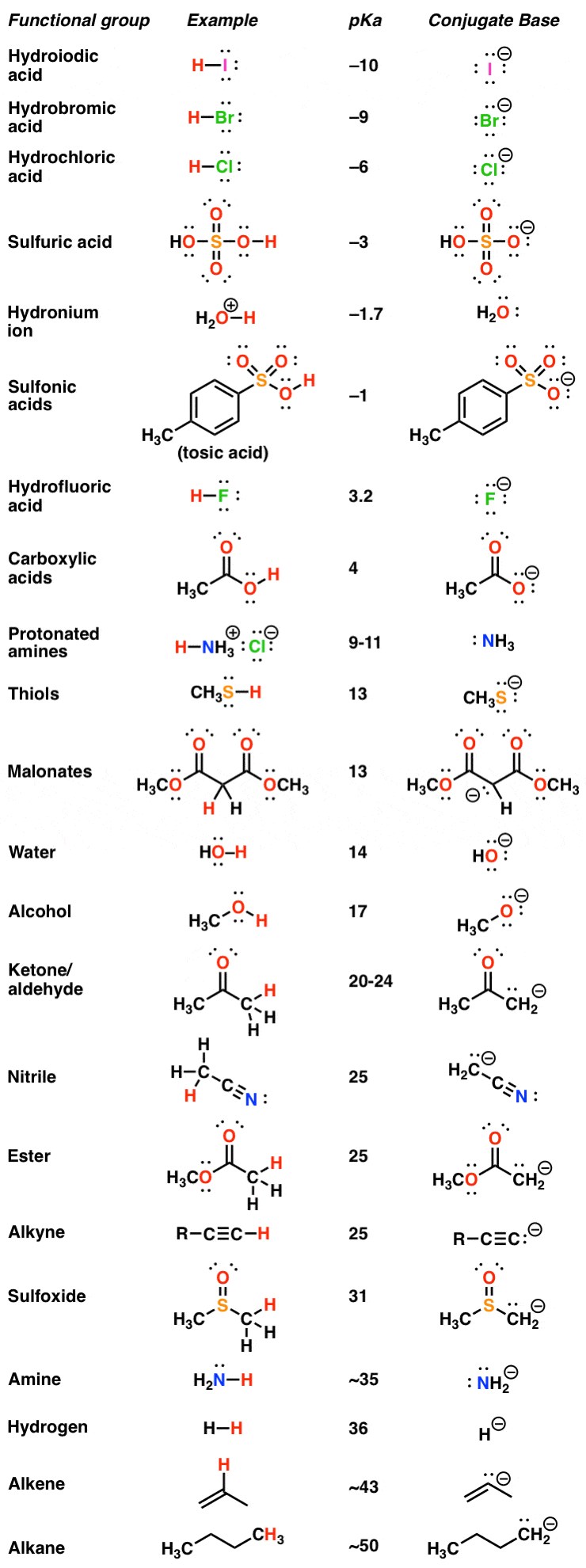
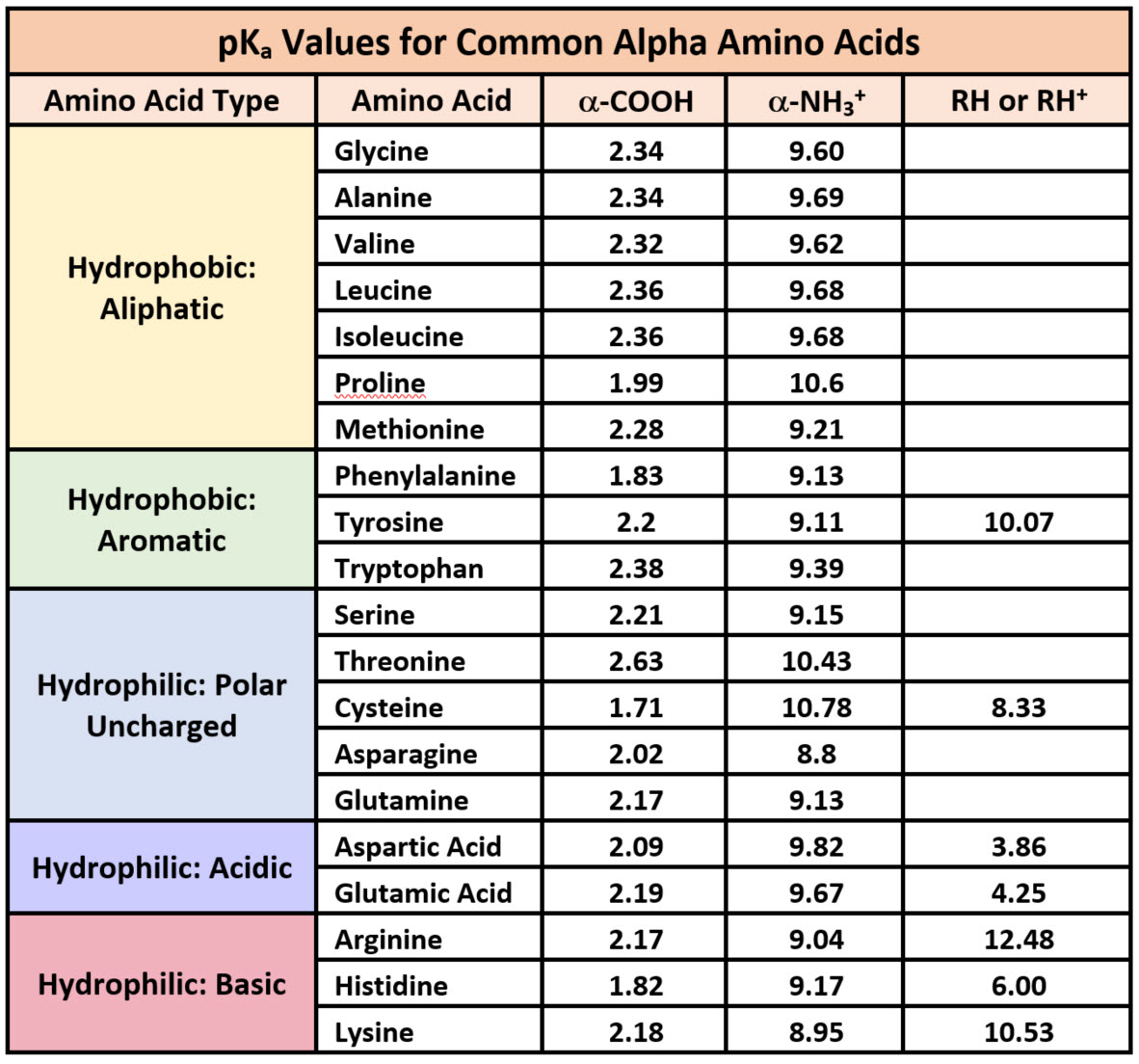
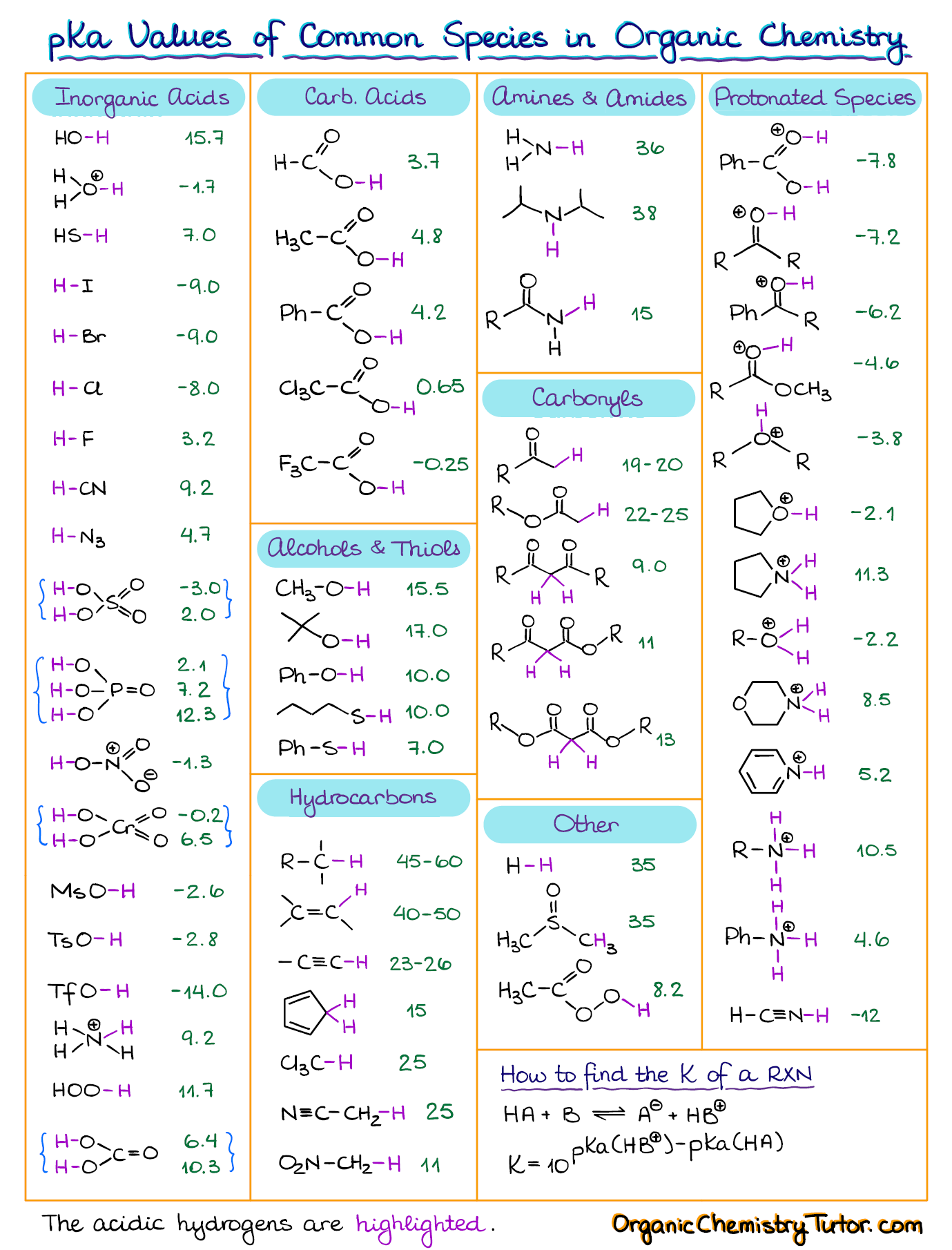
Amino Acid Chart With Pka
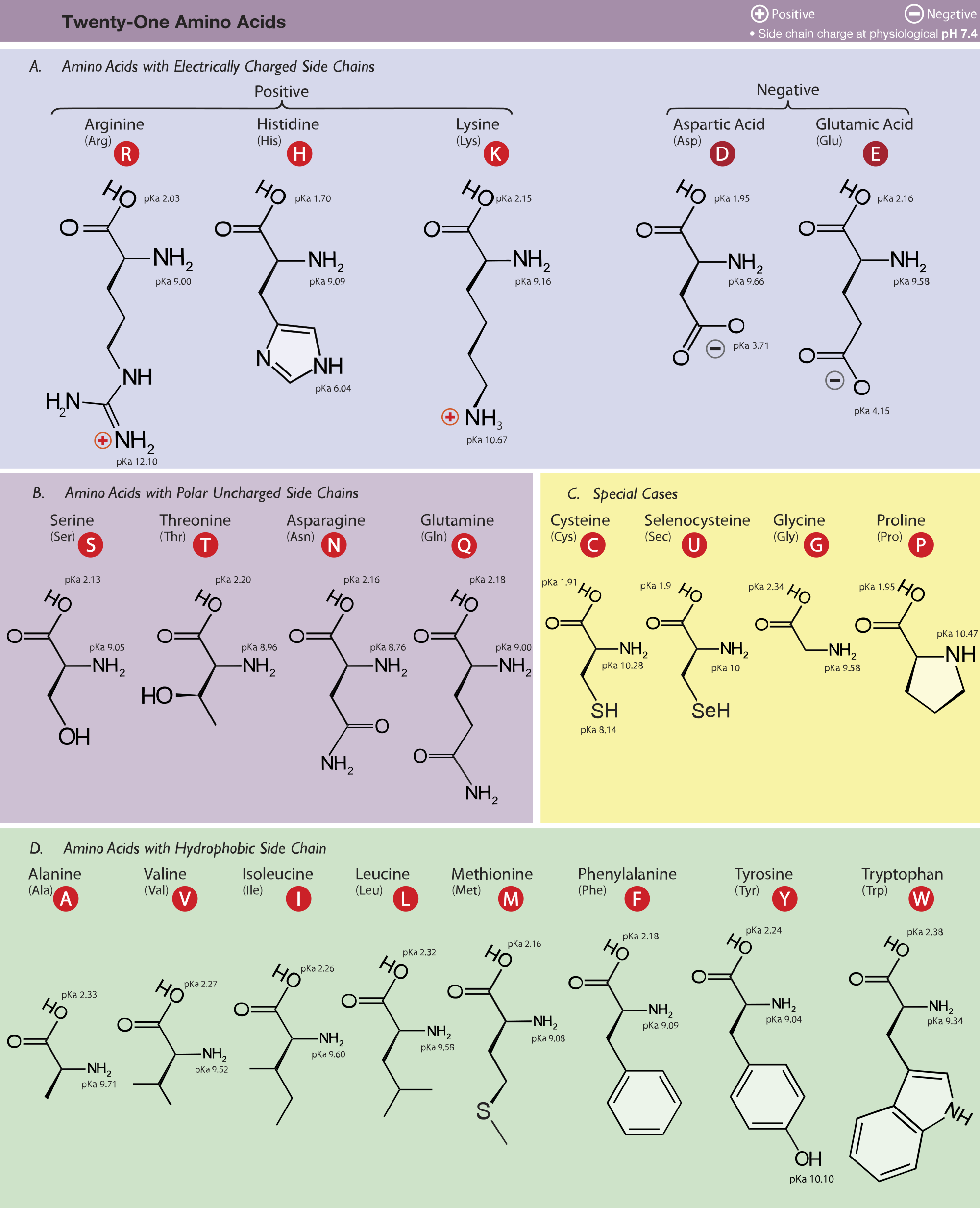
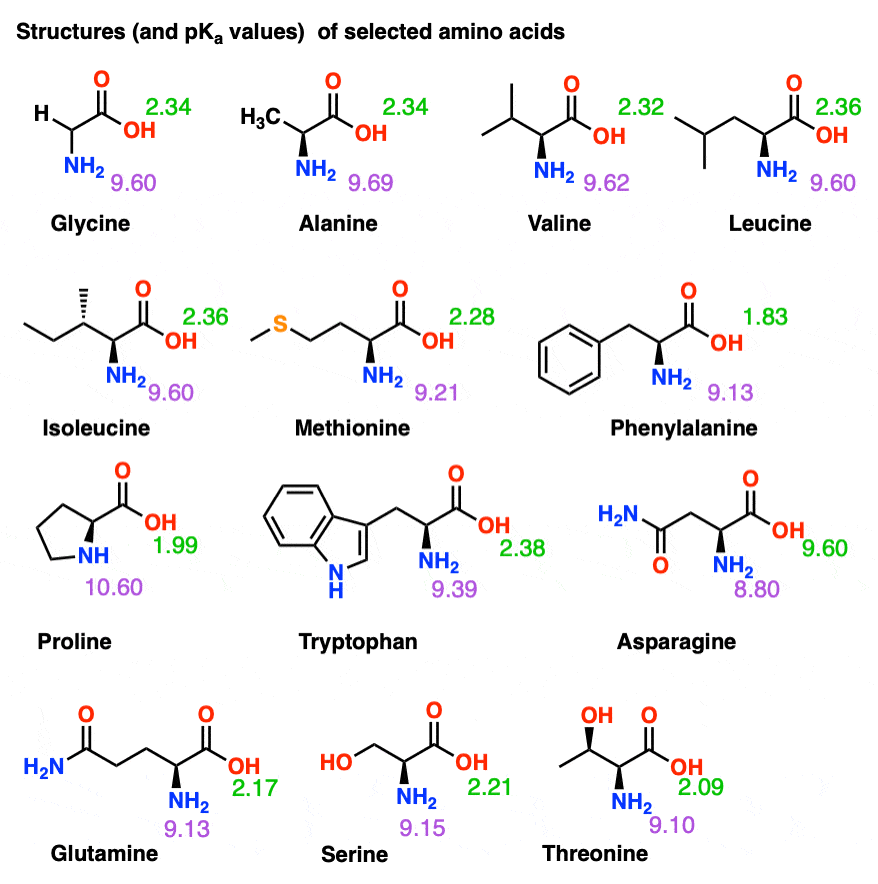
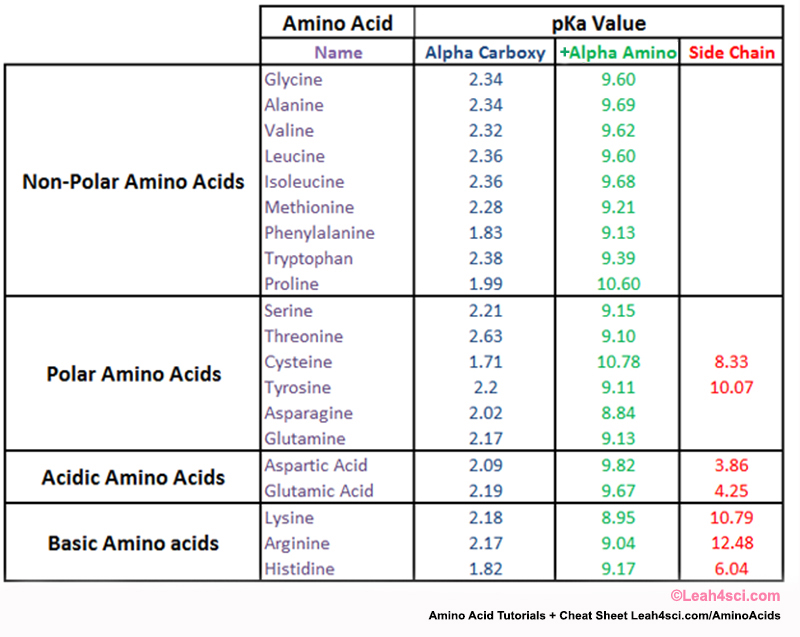
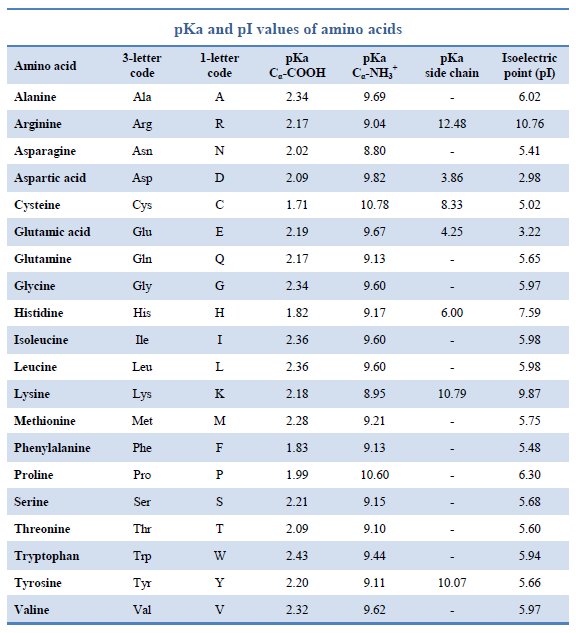
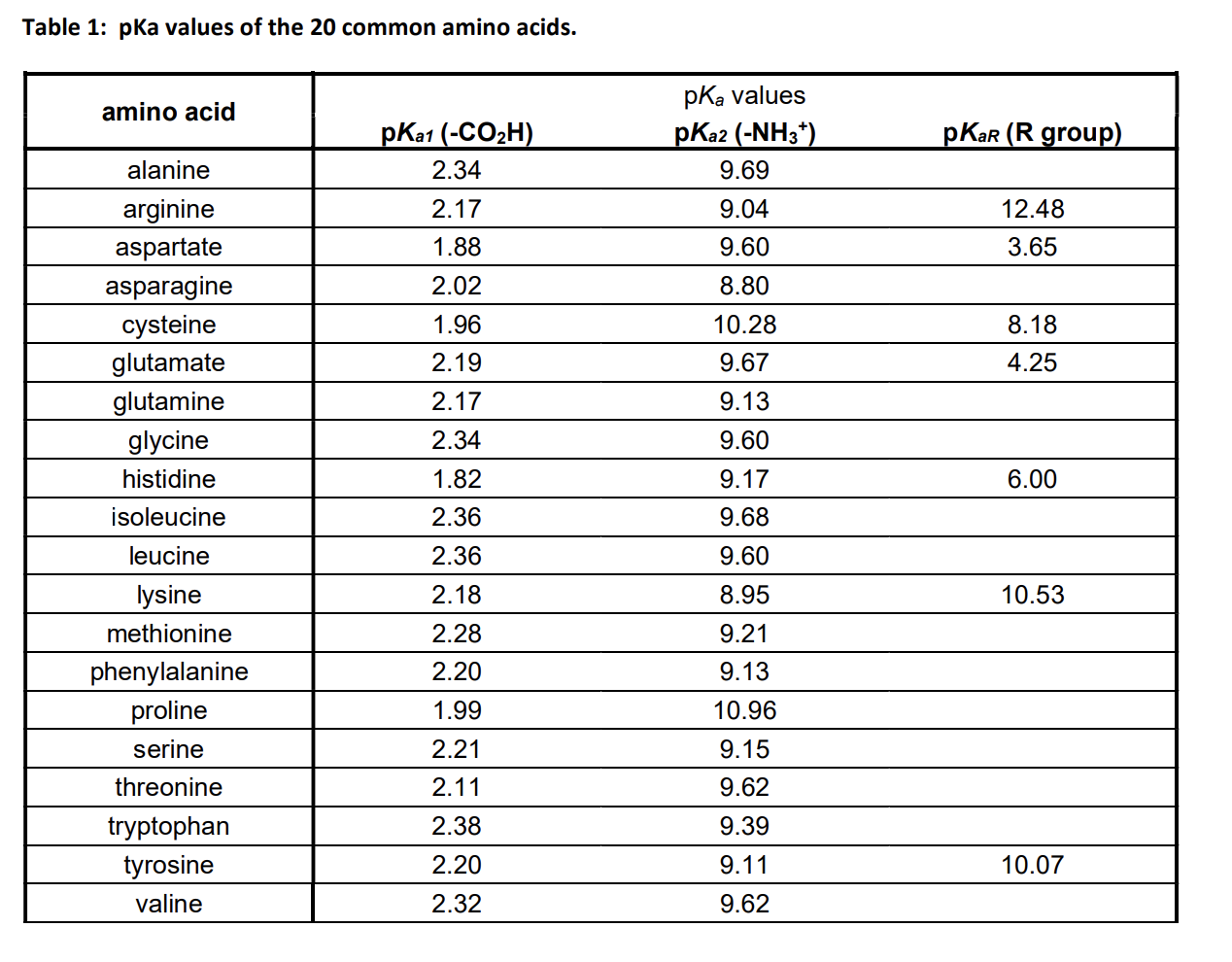
Amino Acid Chart With Pka - Refer to the charts and structures below to explore amino acid properties, types, applications, and availability. Calculating isoelectric point pi from pka values. Titration curves show the neutralization of these acids by added base, and the change in ph during the titration. Web karen steward, phd. Web for amino acids with only one ionizable acidic group (carboxyl group) and one ionizable basic group (amino group), the pi can be calculated as the average of the two pka values. That is a daunting task for 20 amino acids. Amino acid pka c pka n pka r pi; Web most biochemistry courses will require you to know the following: Pi calculations for amino acids with acidic side chains. Web using a pka table. Calculating isoelectric point pi from pka values. Amino acids are the building blocks that form polypeptides and ultimately proteins. Web table of pk a and pi values. Amino acids are the building blocks of proteins. Web the pka is a measure of the strength of an acid, i.e., the lower the pk a stronger the acid. Register for free to listen to this article. Conjugate acid of −nh2 − nh 2, i.e., −nh+3 − nh 3 + has pk a ~10. At neutral ph the amino group is protonated, and the carboxyl group is deprotonated. Refer to chart below to explore structures, properties and types for each of the 20 standard amino acids. Web amino acid structure (video) | khan academy. Web for amino acids with only one ionizable acidic group (carboxyl group) and one ionizable basic group (amino group), the pi can be calculated as the average of the two pka values. At neutral ph the amino group is protonated, and the carboxyl group is deprotonated. Titration curves show the neutralization of these acids by added base, and the change. Web table \(\pageindex{2}\) shows the standard pk a values for the amino acids and can be used to predict the ionization/charge status of amino acids and their resulting peptides/proteins. Conjugate acid of −nh2 − nh 2, i.e., −nh+3 − nh 3 + has pk a ~10. For the four amino acids with either a strongly or weakly acidic side chain,. Web table of pk a and pi values. Amino acids are the building blocks that form polypeptides and ultimately proteins. Web amino acid structure (video) | khan academy. For example, let's consider alanine (ala) again. At neutral ph the amino group is protonated, and the carboxyl group is deprotonated. They contain an amino group, carboxylic acid group, alpha carbon, and side chain. Calculating isoelectric point pi from pka values. Web table of pk a and pi values. For example, let's consider alanine (ala) again. Amino acids are the building blocks that form polypeptides and ultimately proteins. Glycine alanine valine leucine isoleucine proline serine threonine cysteine methionine asparagine glutamine phenylalanine tyrosine tryptophan lysine arginine histidine aspartate glutamate 2.4 2.4 2.3 2.3 2.3 2.0 2.2 2.1 1.9 2.1 2.1 2.2 2.2 2.2 2.5 2.2 1.8 1.8 2.0 Summary of pkas of amino acids The chemical nature of this side chain determines the unique properties of each amino acid.. Calculating isoelectric point pi from pka values. Web table of contents. Web all amino acids have the same basic structure, which is shown in figure 2.1. Amino acids are the building blocks of proteins. How to use a pka table to determine relative acid strengths. That is a daunting task for 20 amino acids. Web the pka is a measure of the strength of an acid, i.e., the lower the pk a stronger the acid. Pi calculations for amino acids with acidic side chains. For the 13 amino acids with a neutral side chain, pi is the average of pk a1 and pk a2. How. Most amino acids have a chiral carbon, which allows them to rotate polarized light. Refer to the charts and structures below to explore amino acid properties, types, applications, and availability. Pi calculations for amino acids with basic side chains. Web most biochemistry courses will require you to know the following: Amino acids are the building blocks that form polypeptides and. Summary of pkas of amino acids Web the r group for each of the amino acids will differ in structure, electrical charge, and polarity. The isoelectric points range from 5.5 to 6.2. Web for amino acids with only one ionizable acidic group (carboxyl group) and one ionizable basic group (amino group), the pi can be calculated as the average of. Web the pka is a measure of the strength of an acid, i.e., the lower the pk a stronger the acid. Refer to chart below to explore structures, properties and types for each of the 20 standard amino acids. Amino acid pka c pka n pka r pi; At neutral ph the amino group is protonated, and the carboxyl group. Web the r group, which differs for each amino acid, will determine its structure, polarity and ph. Web for amino acids with only one ionizable acidic group (carboxyl group) and one ionizable basic group (amino group), the pi can be calculated as the average of the two pka values. For example, the pi of the amino acid glycine is around 5.97, while the pi of lysine, with a basic side chain, is approximately 9.74. Register for free to listen to this article. Most amino acids have a chiral carbon, which allows them to rotate polarized light. For example, let's consider alanine (ala) again. Web table of pk a and pi values. Web using a pka table. Thus, amino acids with (chemically) similar side groups can be expected to function in similar ways, for example, during protein folding. Want to join the conversation? The isoelectric point, pi, is the ph at which negative and positive charges are balanced. Titration curves show the neutralization of these acids by added base, and the change in ph during the titration. At neutral ph the amino group is protonated, and the carboxyl group is deprotonated. Glycine alanine valine leucine isoleucine proline serine threonine cysteine methionine asparagine glutamine phenylalanine tyrosine tryptophan lysine arginine histidine aspartate glutamate 2.4 2.4 2.3 2.3 2.3 2.0 2.2 2.1 1.9 2.1 2.1 2.2 2.2 2.2 2.5 2.2 1.8 1.8 2.0 Calculating isoelectric point pi from pka values. Web table of contents.pKa Table and How to Use It — Organic Chemistry Tutor
Amino Acid Chart Pka
3.1 Amino Acids and Peptides Biology LibreTexts
AcidBase Equilibrium Part 1 How to Use the pKa Table — Organic
Amino Acid Study Guide Structure and Function Albert.io
Amino Acids And Pka Values
Amino Acid Charge in Zwitterions and Isoelectric Point MCAT Tutorial
Amino Acid Pka Chart A Visual Reference of Charts Chart Master
Amino acid properties
Amino Acid Chart With Pka
Web Amino Acid Pka And Pi Values.
That Is A Daunting Task For 20 Amino Acids.
Web The Pka Is A Measure Of The Strength Of An Acid, I.e., The Lower The Pk A Stronger The Acid.
Refer To Chart Below To Explore Structures, Properties And Types For Each Of The 20 Standard Amino Acids.
Related Post: